 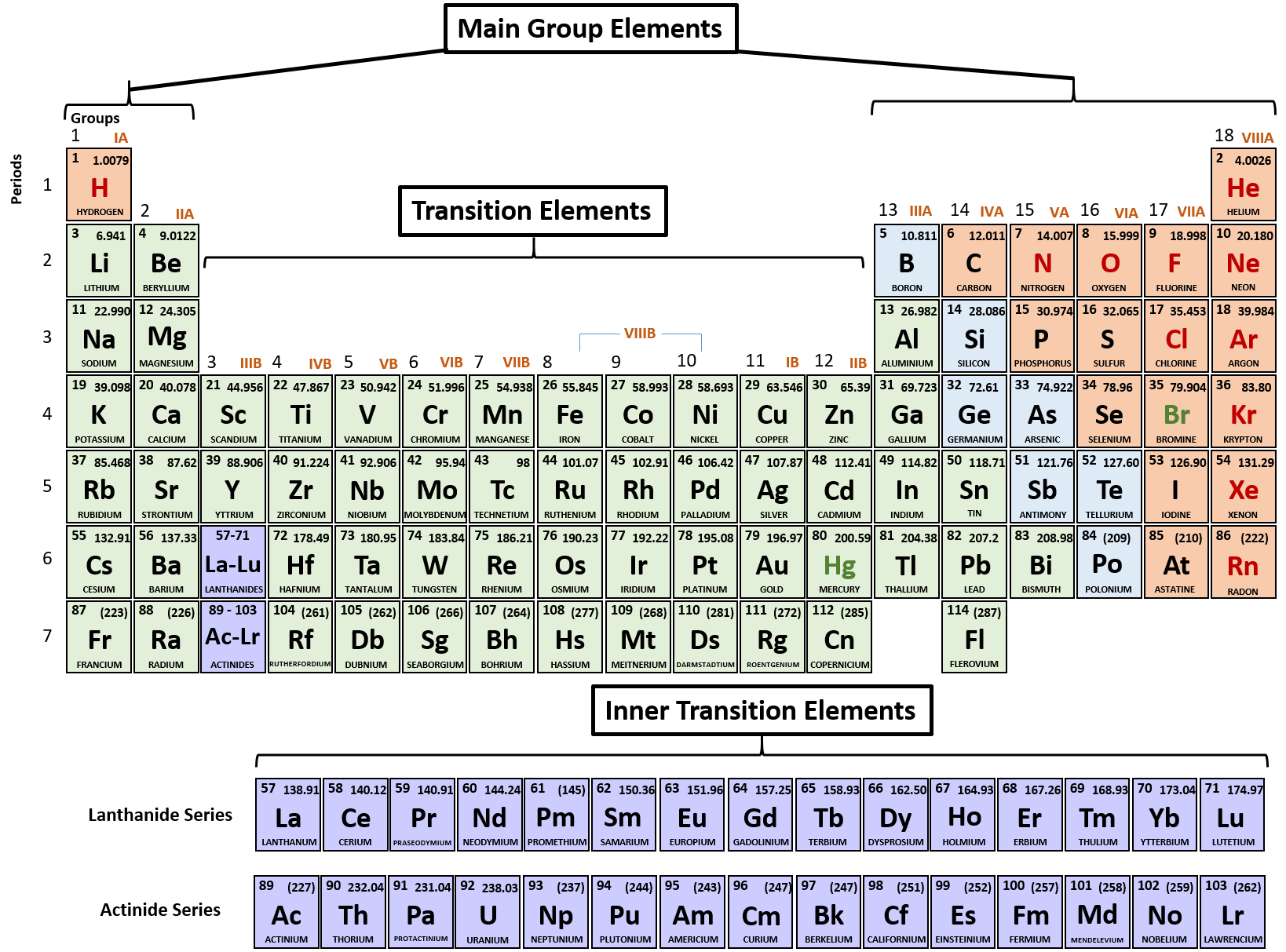
Electrons carry with them electrical energy when they move between atoms. Any electrons that are lost by atoms that are picked up by neutral atoms will turn those neutral atoms into positive atoms.īecause electrons have such ease of movement between atoms, metals are great electricity conductors. For example, gold, silver copper or sodium. What Are Cations?Ĭations are positively charged atoms that are formed from metal atoms. This then results in the formation of cations (positively-charged ions) and, also, the atoms then pick up electrons from each other, which results in the formation of anions (negatively-charged ions). The process of ion formation involves atoms giving up electrons in order to form other atoms. It is precisely this ability that electrons have to move in orbitals while jumping between different atoms is what contributes to the formation of ions. Another thing that makes electrons famous is their free movement around the nucleus in circular directions, making orbital of three dimensions. The isotope of the atom is determined by the number of neutrons and protons therein.Įlectrons are the subatomic particles characterized by their negative charges. Those particles can be neutrons, which are the neutral subatomic particles located in the very center (nucleus) of the atom together with protons with a positive charge. Ion is the name of the subatomic particles that are components of all the atoms. All the metallic elements located on the left part of the Periodic Table have a positive ionic charge, while all the metallic elements located on the right part of the Periodic Table have a negative ionic charge. There is also a very clear way of knowing whether an element has a positive or a negative ionic charge. Even a tiny fleck of it stops time.” - Diane Ackerman “Wonder is the heaviest element on the perioid table. The best way to find out what the ionic charge for a specific element is is by checking the Periodic table. The Period Table With Charges is an essential tool for science students. We can calculate the effective nuclear charge by subtracting the number of inner shell electrons from the number of protons.The elements of the Periodic Table have different ionic charges. How to Calculate Effective Nuclear Charge Because of the varying charge on electrons in different orbitals, we typically refer to the effective nuclear charge, which is the effect of the nucleus experienced by the outermost electron of the atom, taking into account the shielding effect of inner electrons. The presence of electrons on the inner shells of an atom “shield” the outermost electron from feeling the full positive charge. /PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png) 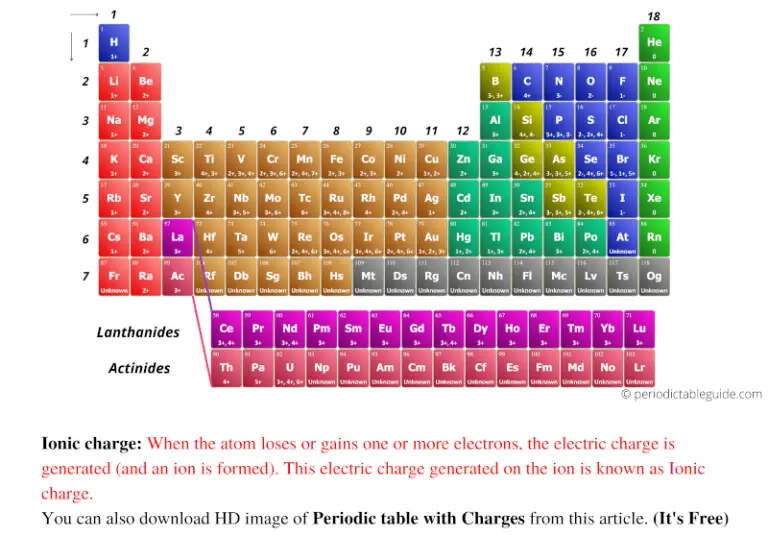
This results in a varying attraction of the nucleus on the electrons surrounding the nucleus, which is known as nuclear charge. However, negatively charged electrons around the nucleus are organized into layers called orbitals which repel each other, and negate some of the positive charge of the nucleus. Electrons are attracted to the nucleus as they are negatively charged. The nucleus of an atom contains positively charged particles called protons. The net attraction on these outer electrons is known as effective nuclear charge. Electrons are attracted to the nucleus as it is positively charged, but electrons in the inner shells can negate some of the attraction of the nucleus on the outermost electrons. Nuclear charge is a measure of the ability of protons in the nucleus to attract the negative electrons in orbit around the nucleus. The equation for calculating nuclear charge is Zeff = Z - S, where Zeff is the effective nuclear charge, Z is the number of protons, and S is the number of inner electrons.ģ. You can calculate effective nuclear charge if you know the number of inner electrons and the number of protons of an atom, both which can be found either from the periodic table or from online resources. How do you calculate effective nuclear charge? Nuclear charge values have been determined for the elements. These values are recorded in encyclopedias, scientific textbooks, and scientific journal articles.Ģ. How do you find effective nuclear charge? These outer electrons are also known as valence electrons.ġ. Elements in different groups on the periodic table have different numbers of electrons in their outermost shells. These negatively charged electrons are arranged into shells which form layers surrounding the nucleus. Refresher: Atoms are composed of a nucleus, containing positively charged protons and neutral neutrons, surrounded by a cloud of negatively charged electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
